The Unseen Guardian: Elevating Precision and Purity with Advanced Pipette Filter Technology
In the intricate landscape of modern scientific research, diagnostics, and pharmaceutical development, the integrity of samples and the accuracy of measurements are paramount. Every step, from sample preparation to final analysis, carries the potential for contamination that can compromise results, invalidate studies, and incur significant financial losses. At the heart of maintaining sample purity and protecting sensitive laboratory equipment lies a crucial, yet often overlooked, component: the pipette filter.
These micro-porous barriers, typically engineered from advanced polymer materials like High-Density Polyethylene (HDPE) or Ultra-High Molecular Weight Polyethylene (UHMW-PE), are indispensable for preventing aerosol cross-contamination, safeguarding pipette mechanisms from corrosive liquids, and ensuring the reliability of laboratory workflows. This comprehensive article delves into the critical role, technical specifications, manufacturing intricacies, and diverse applications of these essential filtration devices, providing a deep dive for B2B decision-makers and technical professionals seeking unparalleled quality and performance in their laboratory consumables.
The Critical Role of Pipette Filters in Modern Laboratories
The demand for pristine laboratory environments and uncontaminated samples has never been higher. Factors such as increasing sensitivity of analytical techniques (e.g., PCR, mass spectrometry), the rise of high-throughput automated systems, and stringent regulatory requirements (e.g., FDA, GLP) amplify the need for robust contamination control. This is where effective pipette filtration becomes non-negotiable.
Preventing Aerosol Cross-Contamination
During aspiration and dispensing, pipettes generate aerosols—microscopic liquid particles that can carry DNA, RNA, proteins, or microbial contaminants. These aerosols can travel into the pipette shaft, contaminating subsequent samples or even the pipette itself, leading to false positives, false negatives, and unreliable experimental data. A high-quality pipette filter acts as an impenetrable barrier, blocking these aerosols before they can reach the delicate internal components of the pipette, thereby preserving sample integrity across experiments.
Protecting Pipette Mechanisms and Extending Lifespan
Beyond biological contamination, pipettes are frequently exposed to corrosive chemicals, volatile organic compounds, and radioactive materials. Without a protective barrier, these substances can enter the pipette body, causing mechanical damage, corrosion, or contamination that requires costly repairs or premature replacement. The hydrophobic nature of most pipette filter materials prevents liquid from entering the pipette, significantly extending the instrument’s operational life and maintaining its calibration accuracy.
Ensuring Reproducibility and Compliance
In regulated environments, every effort must be made to minimize variables and ensure reproducible results. The consistent performance of a pipette filter is a key factor in achieving this. By eliminating a major source of contamination, filters contribute directly to data reliability, critical for regulatory submissions, clinical diagnostics, and drug development where deviations can have severe consequences.
Understanding the Technology: Materials and Design of Pipette Filters
The effectiveness of a pipette filter hinges on the judicious selection of materials and precise engineering of its porous structure. The vast majority of high-performance filters in this category are fabricated from Polyethylene (PE) due to its unique combination of properties.
Porous Polyethylene (PE) as the Material of Choice
Polyethylene, particularly in its High-Density Polyethylene (HDPE) or Ultra-High Molecular Weight Polyethylene (UHMW-PE) forms, is ideal for pipette filtration applications for several reasons:
- Hydrophobicity: PE is inherently hydrophobic, meaning it repels water and aqueous solutions. This property is crucial for blocking liquid aerosols and preventing them from reaching the pipette body, even under conditions of over-pipetting or accidental liquid contact.
- Chemical Inertness: PE exhibits excellent resistance to a wide range of acids, bases, and organic solvents commonly used in laboratories. This ensures that the pe filter itself does not react with samples or introduce extractables, maintaining sample purity.
- Biocompatibility: For life science and medical diagnostic applications, the material must be non-toxic and not interfere with biological samples. Medical-grade PE is rigorously tested for biocompatibility, ensuring it doesn’t inhibit enzyme activity, cell growth, or nucleic acid amplification.
- Porous Structure Control: The manufacturing process of porous PE allows for precise control over pore size and distribution, which directly correlates with filtration efficiency and flow rate. This enables the design of filters optimized for specific applications, from standard liquid handling to highly sensitive molecular biology assays.
- Durability and Mechanical Stability: Despite being porous, pe filters are mechanically robust enough to withstand the forces of aspiration and dispensing without shedding particles or deforming, which is vital for preventing contamination from the filter itself.
Pore Size and Filtration Mechanism
The efficacy of a pe filter is largely defined by its nominal pore size, typically ranging from 0.2 µm to 5 µm. Filters for aerosol blocking often utilize a tortuous path filtration mechanism within a network of interconnected pores. This allows for high particulate retention (including airborne contaminants like bacteria and viral particles) while maintaining a low airflow resistance, crucial for accurate liquid transfer.
The most common pore sizes for general laboratory use are between 1 µm and 5 µm, which are effective against most laboratory aerosols. For highly sensitive applications, such as PCR or cell culture, filters with sub-micron pore sizes (e.g., 0.2 µm or 0.45 µm) may be employed to ensure sterile conditions and prevent the passage of even the smallest contaminants.
The Advanced Manufacturing Process of Pipette Filters
The production of high-performance pipette filters is a highly specialized process, demanding precision engineering, stringent quality control, and often, cleanroom environments to ensure optimal functionality and purity. Unlike heavy industrial components manufactured through casting or forging, these micro-porous polymer components utilize advanced techniques tailored for precise pore structure and material integrity.
1. Material Selection and Preparation
The foundation of a superior pipette filter lies in the selection of medical-grade polyethylene (PE) resins, typically HDPE or UHMW-PE. These virgin polymers are chosen for their consistent molecular weight, purity, and low extractables profile. The raw material arrives in a granular or powder form and undergoes rigorous incoming quality inspection to confirm its chemical composition, density, and flow characteristics meet precise specifications. Any impurities or variations at this stage can compromise the final product’s filtration efficiency or chemical inertness.
2. Porous Structure Formation (Sintering)
The primary method for creating the porous structure of pe filters is a controlled sintering process. In this technique, the PE powder is placed into precision molds and subjected to carefully controlled temperature and pressure. The polymer particles are heated just below their melting point, causing them to fuse together at their contact points while retaining an intricate network of interconnected voids (pores). The key parameters controlled during sintering include:
- Temperature Profile: Precise temperature ramps and holding times ensure optimal bonding without full melting, which would eliminate the porosity.
- Pressure Application: Compaction pressure influences the density and mechanical strength of the final filter.
- Particle Size Distribution: The size and uniformity of the initial PE powder particles significantly determine the resulting pore size and distribution of the pe filter.
- Atmosphere Control: Sintering often occurs in an inert atmosphere to prevent oxidation or degradation of the polymer.
3. Post-Sintering Processing and Shaping
After sintering, the porous PE material is cooled under controlled conditions to prevent warping or stress. The resulting porous “cake” or block is then subjected to precision machining, typically CNC (Computer Numerical Control) machining, to cut and shape the individual pipette filters to their exact required dimensions. This includes creating precise diameters and thicknesses to ensure a snug fit within various pipette tips without compromising airflow or seating stability. This step is critical for consistent performance across different pipette models and tip manufacturers.
4. Stringent Quality Control and Testing
Quality control is integrated throughout the manufacturing process, with particular emphasis on post-production testing. Key inspection standards and tests for a pipette filter include:
- Pore Size Distribution (Porometry): Utilizes techniques like bubble point testing or mercury intrusion porosimetry to verify the average pore size and ensure a tight distribution, critical for consistent filtration efficiency.
- Flow Rate/Air Permeability: Measures the resistance to airflow through the filter. This ensures that the filter does not impede accurate liquid aspiration and dispensing, which would affect pipetting precision.
- Hydrophobicity Test: Verifies the water-repelling properties to confirm aerosol and liquid barrier efficacy.
- Particulate Shedding Test: Ensures the filter itself does not shed micro-particles into the sample path, which would introduce contamination.
- Dimensional Accuracy: CNC measurements and optical inspection ensure each pe filter conforms to tight tolerances for fit and function.
- Biocompatibility and Purity: For medical and life science applications, filters undergo tests for cytotoxicity, pyrogenicity (endotoxins), and the absence of PCR inhibitors or detectable DNA/RNase/DNase. Compliance with standards like ISO 13485 for Medical Devices is crucial.
The entire manufacturing process typically occurs in certified cleanroom environments (e.g., ISO Class 7 or 8) to prevent external contamination during production, ensuring the final product is free from particulates, biological contaminants, and other impurities that could compromise sensitive laboratory work.

Image: Precision Manufacturing of Pipette Filter in a Cleanroom Environment.
The service life of a pipette filter is generally tied to the specific application. In typical laboratory use, a single filter is designed for the lifespan of a pipette tip (single-use), ensuring maximum contamination prevention between samples. However, the inherent durability of the porous PE material means the filter itself, if not exposed to liquid or high aerosol burden, maintains its structural integrity over time. In terms of applicable industries, while the manufacturing processes might echo precision engineering found elsewhere, the end-use of pipette filters is exclusively in scientific, medical, and diagnostic laboratories, including areas like biotechnology, pharmaceuticals, clinical pathology, environmental analysis, and forensic science. Their advantage in these scenarios is primarily through ensuring sample integrity and protecting valuable instrumentation, thereby saving costs related to re-runs and instrument maintenance, which can be viewed as an indirect form of “energy saving” through efficiency.
Technical Specifications and Performance Parameters of a Pipette Filter
Understanding the technical specifications of a pipette filter is crucial for selecting the correct product for specific applications and ensuring optimal laboratory performance. Key parameters dictate the filter’s efficiency, compatibility, and reliability.
Typical Pipette Filter Specifications
| Parameter | Typical Range/Value | Significance for Lab Performance |
|---|---|---|
| Material | Porous HDPE, Porous UHMW-PE | Ensures chemical inertness, biocompatibility, and consistent hydrophobic properties. |
| Pore Size (Nominal) | 0.2 µm, 0.45 µm, 1 µm, 5 µm | Determines filtration efficiency against aerosols (e.g., 0.2 µm for sterile, 5 µm for general protection). |
| Hydrophobicity | Hydrophobic (Water Contact Angle > 120°) | Prevents liquid breakthrough and protects pipette components from aqueous solutions. |
| Air Flow Rate | Typically 10-50 L/min at 10 kPa (for specific dimensions) | Ensures smooth and rapid aspiration/dispensing, maintaining pipetting accuracy. |
| Retention Efficiency | >99.9% for aerosols above nominal pore size | Measures the filter’s ability to block airborne contaminants and cross-contamination effectively. |
| Extractables/Leachables | Non-detectable (DNase/RNase/Human DNA free, PCR inhibitor free) | Guarantees no interference with sensitive molecular assays or analytical tests. |
| Operating Temperature Range | -20°C to +80°C | Ensures stability and performance across various lab conditions, including cold rooms or incubators. |
| Dimensions (Diameter x Length) | Variable, e.g., 2.5 x 4mm, 5 x 7mm, 6.5 x 8mm | Critical for compatibility with specific pipette tips and models (e.g., universal, Rainin, Eppendorf). |
These parameters are not merely numbers; they directly translate into the reliability and integrity of your experimental results. For instance, a filter with a precisely controlled pore size and high retention efficiency is indispensable for PCR applications where even a single aerosolized DNA molecule from a previous sample can lead to false amplification. Similarly, consistent airflow ensures that the pipette maintains its calibrated accuracy, preventing volume discrepancies that could invalidate quantitative assays.
Diverse Applications of Pipette Filters
The ubiquitous nature of pipetting in laboratory settings means that pipette filters find application across a remarkably broad spectrum of scientific disciplines. Their core function of preventing cross-contamination and protecting instrumentation makes them essential in any workflow where sample integrity is critical.
PCR and qPCR
Perhaps the most critical application for filtered pipette tips. PCR (Polymerase Chain Reaction) and qPCR (quantitative PCR) are exquisitely sensitive techniques capable of amplifying even trace amounts of DNA/RNA. Contamination from previous samples (carry-over contamination) via aerosols is a major cause of false positives. Pipette filters with pore sizes typically around 0.2 µm or 0.45 µm provide an effective barrier against such aerosols, safeguarding the integrity of amplification reactions and ensuring reliable diagnostic or research outcomes.
Cell Culture and Sterile Techniques
Maintaining sterility is paramount in cell culture to prevent bacterial, fungal, or mycoplasma contamination that can destroy cell lines or invalidate experiments. Filtered tips are indispensable when working in biological safety cabinets, transferring media, or passaging cells. The pipette filter acts as a physical barrier to airborne microbes, reducing the risk of introducing contaminants into sensitive cell cultures.
Clinical Diagnostics
In clinical laboratories, where patient samples are handled, preventing cross-contamination is not just good practice but a regulatory and ethical imperative. Diagnostic assays for infectious diseases, genetic disorders, or biomarker detection rely heavily on accurate liquid handling. Pipette filters minimize the risk of false results that could lead to misdiagnosis or inappropriate patient management, particularly in high-throughput testing environments.
Drug Discovery and Pharmaceutical Research
From compound screening to formulation development and quality control, pharmaceutical R&D involves handling vast numbers of diverse chemical and biological samples. Pipette filters ensure that precious reagents are not contaminated and that high-cost automated liquid handlers are protected from corrosive or volatile solvents, contributing to the efficiency and integrity of drug development pipelines.
Environmental Testing and Forensic Science
Environmental labs analyze water, soil, and air samples for pollutants, often at trace levels. Forensic labs analyze minute quantities of evidence for DNA or other biomarkers. In both fields, preventing external contamination and ensuring the integrity of the sample during handling is paramount for legally defensible and scientifically sound results. The use of robust pe filters is a standard practice to mitigate contamination risks.
General Laboratory Liquid Handling
Even for routine liquid transfers, the benefits of filtered tips are significant. They provide an added layer of security, protecting both the sample and the pipette from unexpected splashes, over-pipetting incidents, or accidental aspiration of highly viscous or foaming liquids that might otherwise damage the pipette’s internal components.
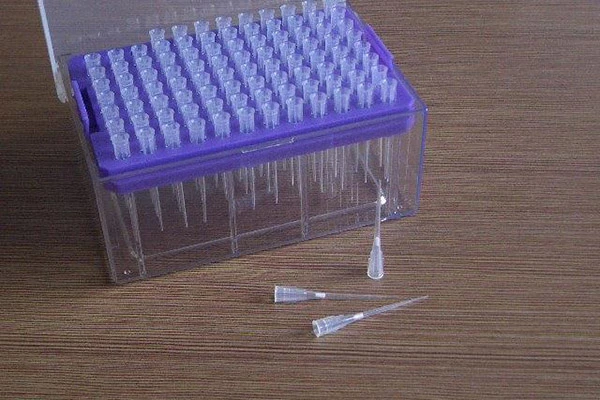
Image: Broad Applications of Pipette Filters Across Research and Diagnostics.
Unpacking the Technical Advantages of Premium Pipette Filters
Investing in high-quality pipette filters goes beyond mere compliance; it directly translates into enhanced data integrity, reduced operational costs, and improved laboratory efficiency. The technical advantages offered by premium filters are multifaceted and profoundly impact the reliability of scientific outcomes.
Superior Contamination Control
The primary advantage is the prevention of both external and cross-contamination. High-performance pe filters are manufactured in certified cleanroom environments, often undergoing irradiation for complete sterility. Their optimized pore structure and hydrophobic properties effectively block aerosols, preventing carry-over between samples and safeguarding sensitive reactions like PCR from ambient particulates, ensuring true-to-sample results.
Enhanced Pipette Longevity and Reduced Maintenance
Pipettes are precision instruments. Exposure to corrosive chemicals, volatile solvents, or even excessive moisture can degrade internal components, leading to decreased accuracy, calibration drift, and premature mechanical failure. The robust hydrophobic barrier of a quality pipette filter acts as a critical shield, protecting plungers, seals, and springs from liquid intrusion. This significantly extends the lifespan of expensive pipettes, reduces the frequency and cost of maintenance, and minimizes instrument downtime.
Reproducible and Reliable Results
In scientific research, reproducibility is the cornerstone of valid findings. By eliminating a major variable—contamination—high-quality pe filters contribute directly to the consistency and reliability of experimental data. This is particularly crucial for studies destined for publication or regulatory submission, where every data point must be traceable and dependable. Reduced re-runs due to contamination also translate to substantial savings in time, labor, and expensive reagents, effectively enhancing laboratory “energy efficiency.”
User Safety and Ergonomics
When handling hazardous biological or chemical agents, filtered tips add a layer of safety for the user. By preventing aerosols from entering the pipette and potentially escaping into the environment, they mitigate exposure risks. Furthermore, well-designed pipette filters ensure consistent airflow, which maintains the pipette’s original ergonomic feel and force requirements, reducing strain on technicians during high-throughput operations.
Broad Compatibility and Versatility
Leading manufacturers offer a range of pipette filters designed to be compatible with a wide array of pipette tips from various brands (e.g., universal, Gilson, Rainin, Eppendorf, Sartorius). This versatility allows laboratories to standardize on high-quality filtration regardless of their existing pipette inventory, simplifying procurement and inventory management. The inherent chemical resistance of PE also ensures compatibility with a vast range of laboratory chemicals, contributing to its “corrosion resistance” in a lab context.
Choosing the Right Pipette Filter: A Manufacturer Comparison
Selecting the optimal pipette filter involves more than just matching pore size; it requires a holistic evaluation of the manufacturer’s capabilities, quality assurance, and service. While the market offers numerous options, discerning buyers focus on long-term value, reliability, and technical support.
Key Factors for Comparison:
- Material Purity and Grade: Is the PE medical-grade? Are certifications (e.g., USP Class VI) available?
- Manufacturing Environment: Are filters produced in a certified cleanroom (e.g., ISO Class 7 or 8)?
- Quality Control Protocols: What tests are performed (pore size, flow rate, extractables)? What are their acceptance criteria?
- Certification and Compliance: Does the manufacturer adhere to relevant standards like ISO 9001, ISO 13485 (for medical devices), and FDA regulations (if applicable)?
- Range of Pore Sizes and Dimensions: Can they supply filters for diverse applications and pipette tip types?
- Customization Capabilities: Do they offer bespoke solutions for unique applications or proprietary tip designs?
- Technical Support and Expertise: Can they provide guidance on filter selection, troubleshooting, and integration?
- Supply Chain Reliability: What are their lead times and capacity for large orders?
- Pricing and Cost-Effectiveness: Balancing upfront cost with long-term benefits (reduced re-runs, extended pipette life).
Manufacturer Comparison Table (Illustrative)
| Feature | Manufacturer A (Our Offering) | Manufacturer B (Competitor Example) | Manufacturer C (Competitor Example) |
|---|---|---|---|
| Material Purity & Grade | Medical-grade UHMW-PE, USP Class VI Certified | Standard HDPE, General Lab Grade | High-grade PE, Limited Certifications |
| Cleanroom Production | ISO Class 7 & 8 Certified Facilities | Partial Cleanroom or Non-Certified | ISO Class 8 Certified |
| Key Certifications | ISO 9001, ISO 13485, FDA-DMR Compliant | ISO 9001 Only | ISO 9001, CE Mark |
| Customization Offered | Extensive (Pore Size, Dimensions, Shape) | Limited (Standard Sizes Only) | Moderate (Some Dimensional Variations) |
| Quality Assurance Testing | 100% Optical Inspection, Batch Porometry, Flow, Extractables, Biocompatibility | Batch Sampling for Porometry & Flow | Automated Dimensional Check, Random Flow Test |
| Technical Support | Dedicated Application Engineers, Fast Response | General Customer Service | Online FAQ & Email Support |
| Typical Lead Time (Large Orders) | 4-6 Weeks (Negotiable for High Volume) | 8-12 Weeks | 6-10 Weeks |
This comparison highlights the differentiators in the market. While some manufacturers prioritize cost-effectiveness through streamlined production, leading suppliers distinguish themselves through rigorous material selection, advanced manufacturing under strict quality systems, and comprehensive post-production testing. Our commitment to ISO 13485 certification, for example, directly reflects a robust quality management system critical for medical device components, underscoring our reliability and commitment to superior pipette filter performance.
Customized Pipette Filter Solutions
The dynamic nature of scientific research and technological advancements often necessitates filtration solutions that go beyond standard off-the-shelf products. Recognizing this, leading manufacturers offer extensive customization capabilities for pipette filters, allowing laboratories and pipette tip manufacturers to tailor solutions precisely to their unique application requirements or proprietary designs.
Tailoring PE Filters for Specific Applications
Customization for a pipette filter can involve several key parameters:
- Custom Pore Sizes: While standard pore sizes suffice for many applications, specific protocols (e.g., highly viscous liquids, specialized aerosol profiles) might benefit from non-standard pore sizes to optimize flow rate versus retention efficiency.
- Unique Dimensions and Shapes: Proprietary pipette tip designs, automated liquid handling platforms with unique tip configurations, or microfluidic devices often require pe filters with specific diameters, lengths, or even custom shapes to ensure a perfect fit and optimal sealing.
- Material Variations: While PE is standard, specific chemical compatibilities or sterilization methods might necessitate variations in polymer grade or even the incorporation of other materials (e.g., PTFE for extreme chemical resistance, though less common for pipette filters).
- Surface Modifications: For highly specialized applications, surface treatments can enhance hydrophobicity or reduce protein binding, further optimizing the filter’s performance for particular sample types.
The Customization Process: A Collaborative Approach
A robust customization program involves a structured, collaborative process to ensure the final product precisely meets client needs:
- Consultation and Requirements Gathering: Initial discussions to understand the application, desired performance criteria (e.g., flow rate, filtration efficiency, chemical compatibility), pipette tip design, and expected volumes.
- Design and Engineering: Our experienced engineers leverage advanced CAD/CAM tools and material science expertise to design the optimal pipette filter geometry and porous structure. This includes selecting the appropriate PE grade and defining the sintering parameters.
- Prototyping and Testing: Rapid prototyping allows for the creation of samples for client evaluation. These prototypes undergo rigorous internal testing for critical parameters (pore size, flow rate, fit) and external validation by the client in their actual application.
- Iteration and Refinement: Based on testing feedback, designs are refined until optimal performance is achieved.
- Scalable Production: Once the design is approved, production is scaled up in our cleanroom facilities, maintaining consistent quality and performance through validated processes.
The benefits of customized pe filters are substantial: optimized performance for niche applications, perfect compatibility with proprietary equipment, enhanced competitive advantage for tip manufacturers, and a stronger alignment between component functionality and overarching scientific goals.
Real-World Impact: Application Case Studies
The theoretical benefits of advanced pipette filter technology are best illustrated through their tangible impact in diverse laboratory environments. These case studies highlight how superior pipette filtration translates into improved data quality, efficiency, and cost savings.
Case Study 1: Mitigating Cross-Contamination in High-Throughput PCR Diagnostics
A large clinical diagnostic laboratory specializing in infectious disease testing faced a persistent challenge with false positives in their high-throughput qPCR assays for respiratory pathogens. Despite rigorous cleanroom protocols and UV sterilization, a low but impactful rate of carry-over contamination was observed, leading to costly re-testing and delayed patient results. After a thorough investigation, aerosolized DNA from positive samples, penetrating standard pipette tips, was identified as a primary culprit. The lab switched to pipette tips integrated with our 0.2 µm pore size pe filters, designed for ultra-high aerosol retention. Within two months, the rate of suspected cross-contamination events dropped by over 85%. This reduction saved the lab an estimated $15,000 per month in reagent costs and labor for re-runs, and significantly improved their diagnostic turnaround time, enhancing patient care.
Case Study 2: Protecting Automated Liquid Handlers in Drug Discovery
A leading pharmaceutical company relied heavily on automated liquid handling robots for high-throughput compound screening. They frequently encountered issues with their pipetting modules, requiring extensive maintenance due to exposure to aggressive DMSO-based solutions and volatile organic solvents. Standard pipette tips offered insufficient protection. Upon adopting tips featuring our chemically resistant, hydrophobic pipette filter (specifically formulated for enhanced solvent resistance), the company observed a dramatic decrease in pipette maintenance interventions. Over a 12-month period, unscheduled downtime for pipette head repairs decreased by 60%, and the lifespan of individual pipetting modules extended by an average of 30%. This translated into significant savings in repair costs and maximized the operational efficiency of their expensive automated systems, accelerating their drug discovery process.

Image: Pipette Filter Integration in Automated Liquid Handling for Enhanced Throughput.
Case Study 3: Ensuring Consistency in Academic Gene Editing Research
A university research group pioneering CRISPR-Cas9 gene editing faced challenges with inconsistent experimental outcomes, often attributing variability to subtle contamination events. Their highly sensitive experiments, involving minute quantities of nucleic acids and enzymes, were extremely vulnerable to cross-contamination. Traditional non-filtered tips were proving inadequate. The group integrated tips featuring our custom-designed, low-binding pe filter, engineered to minimize sample retention while providing robust aerosol protection. This strategic shift resulted in a 25% improvement in their experimental success rate, reducing the number of repeated experiments and optimizing their use of expensive reagents and valuable research time. The increased reproducibility allowed them to accelerate their publication schedule and obtain more definitive results, reinforcing their reputation in the competitive field of genomics.
Ensuring Trust and Reliability: Our Commitment to Quality and Support
For B2B partners, the decision to choose a supplier for critical components like pipette filters extends beyond product specifications. It encompasses the manufacturer’s commitment to quality, adherence to industry standards, and the robustness of their support infrastructure. Our ethos is built on establishing deep trust and demonstrating unwavering reliability.
Authoritativeness through Certifications and Expertise
We stand as an authoritative leader in porous plastic manufacturing, backed by:
- ISO 9001:2015 Certification: Demonstrates our adherence to a globally recognized quality management system, ensuring consistent product quality and customer satisfaction.
- ISO 13485:2016 Certification: Specifically for medical devices, this certification underscores our rigorous controls over the design, development, production, and distribution of medical-grade components, including pipette filters.
- FDA Device Master File (DMF) Support: For partners developing FDA-regulated products, our DMF can streamline your regulatory submissions by providing detailed information on our material and manufacturing processes.
- Decades of Porous Material Expertise: With over two decades in the industry, our R&D investment consistently pushes the boundaries of porous polymer technology, reflected in our innovative pe filters.
- Strategic Partnerships: Collaborations with leading pipette tip manufacturers and instrument developers validate our products’ performance in real-world, high-stakes environments.
Trustworthiness: Quality Assurance, Warranty, and Support
Our commitment to trustworthiness is woven into every aspect of our operations:
- Rigorous Quality Assurance: Every batch of pipette filter undergoes comprehensive testing, including automated optical inspection, porometry, and flow rate verification, to ensure it meets or exceeds specified parameters. Certificates of Analysis (CoA) are available upon request.
- Comprehensive Warranty: We stand behind the quality of our products with a clear warranty against manufacturing defects and performance deviations. Detailed terms are provided with every order.
- Transparent Delivery Cycles: Our supply chain is optimized for efficiency and transparency. For standard products, typical lead times are 4-6 weeks for large orders, with expedited options available. For custom pe filter solutions, lead times are clearly communicated during the quotation phase, factoring in design, prototyping, and validation.
- Dedicated Customer Support: Our team of technical experts and customer service representatives is readily available to assist with product selection, technical inquiries, order tracking, and any post-sales support. We provide detailed documentation, including product specifications and material safety data sheets (MSDS).
- Customer Feedback Integration: We actively solicit and integrate customer feedback into our product development and quality improvement cycles, demonstrating our commitment to continuous enhancement and client satisfaction.
Experience: Client Success Stories
Our long-standing relationships with global leaders in laboratory consumables and medical diagnostics speak volumes about our experience and product efficacy. For instance, a major global distributor of lab supplies reported a 30% increase in customer satisfaction for their filtered pipette tip line after switching to our pipette filters, citing improved consistency and reduced contamination complaints. Another client, a rapidly expanding biotech startup, credited our responsive technical support and custom pe filter solutions for enabling them to bring their novel diagnostic assay to market faster, achieving critical milestones without compromising sample integrity.
Frequently Asked Questions (FAQ) about Pipette Filters
Q1: What is the typical lifespan of a pipette filter?
A1: Pipette filters are designed for single-use, integrated within disposable pipette tips. Their effective lifespan is intended to be for the duration of one liquid transfer or experiment, after which the tip and filter are discarded. This ensures maximum protection against cross-contamination and maintains the integrity of each sample. While the physical material of the pe filter is durable, its performance as an aerosol barrier can be compromised after aspiration of liquids or repeated exposure to high aerosol burdens.
Q2: How do I choose the correct pore size for my pipette filter?
A2: The choice of pore size for a pipette filter depends on the application’s sensitivity and the nature of potential contaminants. For highly sensitive molecular biology applications like PCR, qPCR, or working with viral samples, a smaller pore size (e.g., 0.2 µm or 0.45 µm) is recommended for maximum aerosol and particulate retention, ensuring sterile conditions. For general liquid handling, non-critical assays, or basic pipette protection, larger pore sizes (e.g., 1 µm or 5 µm) may suffice, offering lower airflow resistance while still providing good protection. Consult our technical team for specific recommendations based on your unique workflow.
Q3: Are these filters compatible with all pipette brands and tip manufacturers?
A3: We manufacture a wide range of pipette filters with various dimensions to ensure broad compatibility. Our filters are designed to fit most standard “universal fit” pipette tips, as well as those designed for specific brands like Gilson, Rainin, Eppendorf, Sartorius, and others. We provide detailed dimensional specifications, and we highly recommend testing samples with your specific tips and pipettes to confirm optimal fit and performance. For unique or proprietary tips, we also offer custom manufacturing solutions.
Q4: Can pipette filters prevent chemical contamination of the pipette?
A4: Yes, the inherent hydrophobicity and chemical inertness of the porous PE material in our pe filters provide excellent protection against chemical contamination. The hydrophobic barrier repels aqueous solutions, preventing them from wicking into the pipette shaft. Furthermore, PE’s robust chemical resistance ensures that the filter itself will not degrade or react with most common laboratory chemicals, safeguarding your pipette’s internal components from corrosive or volatile liquids, thus extending its operational life and maintaining its calibration.
Q5: What are the key benefits of using PE filters over filters made from other materials?
A5: Porous Polyethylene (PE) is the material of choice for the majority of high-performance pipette filters due to its unique combination of properties. Key benefits of PE filters include: superior inherent hydrophobicity, excellent chemical resistance to a wide range of lab reagents, high biocompatibility (often certified medical grade), precise control over pore size and distribution during the sintering process, and minimal particulate shedding. While other materials like cellulose or glass fiber might be used in some filtration applications, PE offers the optimal balance of filtration efficiency, pipette protection, and sample integrity for precision liquid handling.
Conclusion
The humble pipette filter, though small in size, plays an indispensable role in safeguarding the integrity of scientific research, diagnostics, and industrial quality control. From preventing insidious aerosol cross-contamination in sensitive molecular assays to protecting expensive automated liquid handlers from corrosive reagents, its impact on data reliability, operational efficiency, and cost reduction is profound. By understanding the advanced manufacturing processes, meticulous quality control, and diverse applications of premium pipette filtration technology, B2B stakeholders can make informed decisions that elevate their laboratory’s precision and purity standards. Choosing a partner with proven expertise, robust quality certifications, and a commitment to customized solutions ensures that your critical liquid handling workflows are not just efficient, but unequivocally reliable.
References
- Clinical and Laboratory Standards Institute (CLSI). (2008). GP29-A: Performance Testing of Pipettes and Disposable Pipette Tips.
- Cappiello, C. M., et al. (2018). Impact of Filtered Pipette Tips on Contamination Control in Molecular Diagnostics. Journal of Clinical Microbiology, 56(10), e00304-18.
- Liu, W., & Wei, L. (2020). Advances in Porous Materials for Biomedical Applications. Advanced Functional Materials, 30(24), 2000570.
- International Organization for Standardization (ISO). (2016). ISO 13485: Medical devices — Quality management systems — Requirements for regulatory purposes.
- Rampf, W. (2010). Pipetting: The Art of Accurate Liquid Handling. American Laboratory, 42(1), 32-34.
- U.S. Pharmacopeia (USP). (2020). USP Class VI Biological Reactivity Tests, In Vivo.
- Heidelberg, P. (2017). Aerosol Barrier Tips – a Must for Every Lab. Eppendorf AG.
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog