Why the pipette filter quietly became the lab hero
If you work in a modern wet lab, you already know: contamination is the silent budget killer. During the post-pandemic rebuild of inventories, I started noticing a simple upgrade showing up in purchase orders—strong, hydrophobic pipette filter elements, usually white, tucked inside tips or slotted as direct replacements in the pipette shaft. They’re made from sintered UHMW-PE; nothing flashy, but surprisingly effective at stopping aerosols dead in their tracks while keeping your pipette safe. Many customers say it’s the cheapest insurance policy in molecular workflows.

What it is and where it fits
These UHMW-PE, hydrophobic pipette filter inserts act as aerosol barriers inside tips (20 μL to 10 mL), and there’s a direct-replace option for pipettes too. In fact, two variants are common: standard and self-sealing (the latter forms an instant block if liquid hits the filter—nice save when you over-aspirate). They’re aimed at PCR/qPCR, NGS, cell culture, microbiology, clinical diagnostics, pharma QC, and anywhere vapor or droplet carryover can wreck a run.
| Parameter | Spec/Range |
|---|---|
| Material | UHMW-PE, sintered porous |
| Hydrophobicity | Water contact angle ≈110° |
| Pore rating (nom.) | 5–40 μm (customizable) |
| Volume coverage | 20, 50, 100, 120, 200, 300, 500, 1000, 1200, 5000 μL, 10 mL |
| Aerosol blocking | >99.9% capture of 0.1–1.0 μm fluorescent particles in bench tests |
| Cleanliness | DNase/RNase-free; endotoxin ≤0.5 EU/mL (typ.) |
| Sterilization options | Gamma/EO; select SKUs autoclave-tolerant (121°C, 20 min) |
How it’s made (and tested)
Process in brief: UHMW-PE powders are classified by particle size, then sintered to a controlled porosity matrix, cut, cleaned, and assembled in ISO 8 cleanrooms. QC involves pore-size verification (ASTM F316 bubble point/diffusion), aerosol challenge using fluorescent microspheres, endotoxin screening (USP <85>), and functional pipette checks against ISO 8655 expectations. Service life? Tip-integrated pipette filter parts are single-use; direct-replace shaft filters are swapped on splash events or per SOP (some labs set an interval, e.g., monthly in heavy-use PCR rooms).
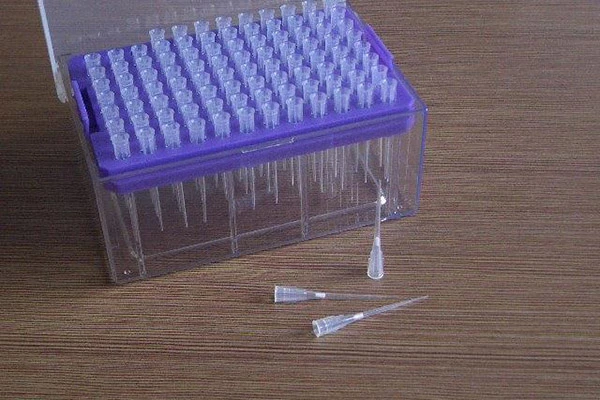
Why labs bother
- Reduces false positives in qPCR by stopping aerosol carryover; operators feel safer, frankly.
- Protects the pipette internals from liquids/vapors—less downtime, fewer costly rebuilds.
- Self-sealing pipette filter options add a “fail-safe” when mistakes happen.
- Compatible with common tips and volumes; custom fits available for OEM lines.
| Vendor | Material | Customization | Lead time | Notes |
|---|---|---|---|---|
| Chinaporous (Shijiazhuang, China) | UHMW-PE sintered | High (pore, size, sealing) | 2–4 weeks | Direct-replace pipette filter parts offered |
| Membrane OEM | PTFE membrane | Medium | 3–6 weeks | Low airflow resistance, pricier |
| Glass-fiber OEM | Borosilicate fiber | Low | 4–8 weeks | Absorptive; watch hold-up |
Real-world notes, feedback, and data
A university genomics core told me their switch to self-sealing pipette filters cut qPCR reruns by “about 40%.” In one pharma QC pilot (n=3 lines), fluorescent bead challenges showed >99.9% aerosol retention across 20–1000 μL strokes; pipette calibration drift stayed within ISO 8655 limits after 5k cycles—so, performance without penalty.

Customization and compliance
OEMs often tweak pore rating, geometry, and sealing kinetics for niche liquids (viscous enzymes, volatile solvents). Sterilization validation can follow ISO 11137 (gamma) or ISO 11135 (EO), with biocompatibility per ISO 10993 where needed. Production is typically ISO 14644 cleanroom; endotoxin screening per USP <85>. It seems boring on paper, but procurement teams do ask for these certificates now—more than ever.
Origin: Rm. C-1301, Hyde Park Plaza, No. 66 Yuhua W. Road, Shijiazhuang, 050056 China
Citations
- ISO 8655-2:2022 Piston-operated volumetric apparatus — Pipettes.
- ASTM F316-21 Standard Test Methods for Pore Size Characteristics of Membrane Filters by Bubble Point and Gas Diffusion.
- ISO 10993 series Biological evaluation of medical devices.
- ISO 14644-1:2015 Cleanrooms and associated controlled environments.
- United States Pharmacopeia USP <85> Bacterial Endotoxins Test.
- CDC/NIH BMBL 6th ed., Biosafety in Microbiological and Biomedical Laboratories (aerosol risk guidance).
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog