Let’s talk about the humble pipette filter—the tiny barrier that quietly saves PCR runs, protects operators, and keeps pipettes alive longer than they probably deserve. I’ve seen labs try to cut corners here; to be honest, it never ends well. Aerosols travel. Contamination sneaks in. And then the data looks… suspicious.

What’s trending in lab filtration
In fact, three things are driving adoption: cleaner PCR/NGS workflows, stricter QA in pharma QC, and biosafety culture. Hydrophobic, UHMW-PE sintered elements—like the ones here—are the quiet stars. They form aerosol barriers without messing with pipetting accuracy. Many customers say their false-positive rates drop after switching to a pipette filter, and I’ve heard the same from core facilities, especially during high-throughput seasons.
Materials and manufacturing (quick, but technical)
- Material: UHMW-PE, precision sintered for controlled pore structure; inherently hydrophobic.
- Form factors: standard and self-sealing types (the latter closes on over-aspiration to block liquid).
- Methods: resin selection → sintering → die-cutting/micro-punching → tip integration → cleanroom packaging.
- Testing: bubble point/air permeability (ASTM F316), pipette system accuracy (ISO 8655), particle control (ISO 14644), bioburden as applicable.
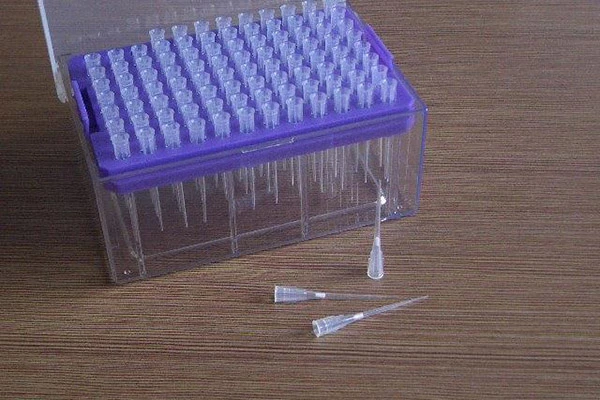
Product specs at a glance
Available capacities: 20 μl, 50 μl, 100 μl, 120 μl, 200 μl, 300 μl, 500 μl, 1000 μl, 1200 μl, 5000 μl, and 10 ml. Direct-replacement formats fit popular tip lines. A pipette filter like this blocks liquids and vapors from reaching internal pistons—yes, it protects both your sample and the instrument.
| Parameter | Typical Value | Notes |
|---|---|---|
| Material | UHMW-PE (sintered) | Hydrophobic; low extractables |
| Pore size | ≈ 5–50 μm | Customized for airflow vs. barrier |
| Aerosol barrier | ≥ 99% in challenge tests | Real-world use may vary |
| Operating temp | -20 to 80 °C | Short-term excursions ok |
| Sterility | Non-sterile or EO-sterile | Lot C of sterile available |
Applications and industries
- Genomics and PCR/NGS: lower carryover and false positives.
- Cell culture and virology: biosafety-first pipetting; pipette filter reduces aerosol drift.
- Pharma QC/QA and compounding (USP <797> environments).
- Forensics and environmental testing—where trace is everything.
Customization and lifecycle
Options include pore size, diameter/height, self-sealing chemistry, and packaging (bulk vs. racked). Service life is single-use per tip; shelf life typically 3–5 years sealed. Origin: Rm. C-1301, Hyde Park Plaza, No. 66 Yuhua W. Road, Shijiazhuang, 050056 China. Certifications often requested: ISO 9001/13485, RoHS/REACH compliance. A good pipette filter should ship with batch CoA and permeability data.

Vendor snapshot (realistic, not hype)
| Vendor | Strengths | Lead Time | MOQ | Certs |
|---|---|---|---|---|
| Porous OEM (China) | Custom UHMW-PE, self-sealing options, direct-fit filters | ≈ 2–4 weeks | Low–mid | ISO 9001/13485, RoHS/REACH |
| Global Brand A | Broad tip ecosystem, global distribution | Stock–2 weeks | Mid | ISO 13485, cGMP support |
| Economy Supplier B | Value pricing for routine use | 3–6 weeks | High | ISO 9001 |
Field notes and a quick case
One genomics lab reported that after moving to self-sealing pipette filter tips for a high-throughput SARS-CoV-2 PCR panel, cross-contamination incidents dropped from ~1.8% to 0.1% over 8 weeks (n≈12,000 samples). Their pipette service intervals stretched, too—surprisingly by almost a quarter. That aligns with what we hear: fewer liquid incursions, fewer repairs.
Compliance and test data you should ask for
- Per ISO 8655, verify accuracy/precision with the filter installed.
- Bubble point/permeability curves (ASTM F316) and aerosol challenge method summary.
- Cleanroom/particulate control declarations (ISO 14644) and, where applicable, USP <797> alignment.
- RoHS/REACH declarations for materials—always good to have on file.
Authoritative references:
- ISO 8655:2022 Piston-operated volumetric apparatus.
- USP <797> Pharmaceutical Compounding—Sterile Preparations.
- CDC/NIH Biosafety in Microbiological and Biomedical Laboratories (BMBL), 6th ed.
- ASTM F316 Standard Test Methods for Pore Size Characteristics of Membrane Filters.
- ISO 14644-1 Cleanrooms and associated controlled environments.
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog