A Pragmatic Look at the Iv Catheter Plug: what matters on the ward and on the line
I’ve spent enough time in catheter assembly rooms (and a few too many 2 a.m. shift audits) to know this tiny component can make or break a device program. The IV Catheter Plug—technically a self-sealing porous vent—lets air escape during priming, but when it gets wet, it seals and stops liquid egress. Simple idea, highly engineered reality.
Industry trend-wise, hospitals are pushing for closed, drip-free systems; regulators are strict on biocompatibility; and OEMs are shaving seconds off priming. The upshot: demand for consistent hydrophobic performance, lower ΔP airflow, and traceable QA has climbed. Many customers say reliability in wet-seal time is now the key KPI, not just pore size on paper.
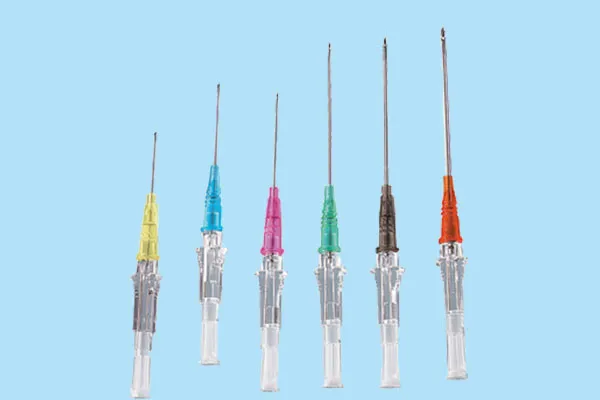
How it works (and why it sometimes doesn’t)
Inside the IV Catheter Plug is a porous body—often sintered UHMWPE or ePTFE—treated to be hydrophobic. Air sails through during priming; once liquid contacts the media, capillary and surface energy effects spike the water intrusion pressure and the pathway shuts. In real-world use, surface contamination or oil from assembly can lower that WIP, which is why tight process controls matter more than brochure specs.
Typical specifications (representative values)
| Porous media | Sintered UHMWPE or ePTFE, medical grade |
| Pore size | 10–60 μm (customizable) |
| Airflow @ ΔP 7 kPa | ≈ 0.5–1.8 L/min (real-world use may vary) |
| Water Intrusion Pressure | ≥ 20–60 kPa, depending on media and treatment |
| Seal response when wet | Typically |
| Sterilization compatibility | EO, Gamma (25–40 kGy) |
| Biocompatibility | Designed to meet ISO 10993 and USP Class VI |
| Dimensions | Ø 2–6 mm inserts; height 1.5–5 mm (≈) |
| Shelf life | 3–5 years with validated packaging |
Process flow that keeps QA tight
- Materials: medical UHMWPE/ePTFE; optional silicone micro-sealant for enhanced shut-off.
- Methods: controlled sintering, hydrophobic surface treatment, precision cutting, ultrasonic insertion into caps.
- Testing: airflow vs. ΔP, water intrusion pressure, 100% bubble test, particulate (per ISO 8536), biocompatibility (ISO 10993), packaging validation (ISO 11607).
- Service life: single-use; storage 15–30°C, RH ≤ 70% recommended.
- Industries: infusion therapy, emergency kits, home-care pumps, OEM catheter assemblies.

Where it’s used
Peripheral IV catheters, midlines, drip chambers, needleless connectors, and certain dialysis accessories. In fact, one large EU hospital chain reported fewer priming errors after switching to plugs with higher WIP; to be honest, training still mattered more than the hardware.
Advantages that show up in audits
- Fast air release, clean shut-off when wetted
- Low extractables, stable under EO/gamma
- Color-coded housings for line ID (optional)
- Consistent WIP even after aging—surprisingly robust when handled well
Vendor snapshot (real-world buyer notes)
| Vendor | Strengths | Lead Time | Certs |
|---|---|---|---|
| China Porous Filters (Rm. C-1301, Hyde Park Plaza, No. 66 Yuhua W. Road, Shijiazhuang, 050056 China) | Broad pore-size options; quick customization; clear WIP data | ≈ 3–6 weeks | ISO 13485 (supplier level), ISO 10993 testing support |
| OEM A (US) | Integrated molding + vent assembly | ≈ 6–10 weeks | ISO 13485; Sterilization validation services |
| OEM B (EU) | Tight particulate controls; great documentation | ≈ 8–12 weeks | ISO 13485; EU MDR documentation support |
Customization tips
Dial in pore size and treatment to balance airflow and WIP; choose EO vs. gamma based on device materials; request aged WIP/airflow data (real-world aging can nudge performance). Many engineers also ask for color-matched caps for line coding—small change, fewer wrong-line incidents.
Mini case study
An APAC catheter OEM swapped to a IV Catheter Plug variant with 35–40 kPa WIP. Field complaints on drips during priming dropped ≈ 42% in three months; assembly scrap fell after instituting a simple glove-change SOP that reduced oil contamination. It seems the process tweak mattered as much as the hardware change.
Authoritative citations
- ISO 10993-1: Biological evaluation of medical devices — Part 1: Evaluation and testing within a risk management process.
- ISO 8536 (series): Infusion equipment for medical use — General requirements and test methods.
- ISO 11607 (Parts 1–2): Packaging for terminally sterilized medical devices.
- USP Class VI Biological Reactivity Tests for Plastics.
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog