Inside the hardware of safer infusions: the Iv Catheter Plug
If you’ve ever wondered why modern IV starts feel smoother and—frankly—less messy than they used to, there’s a tiny hero hiding in the hub. The Iv Catheter Plug is a self-sealing vent that lets air escape during priming, then shuts tight the moment it gets wet. Simple idea, smart execution. And yes, it’s become a quiet trend across infusion devices this past decade.
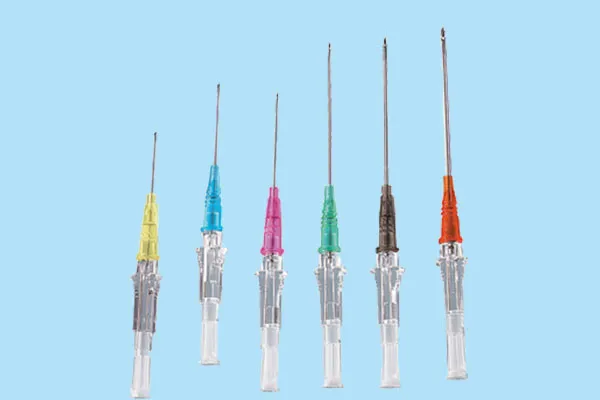
What it is and how it works
In plain terms, a Iv Catheter Plug is a porous polymer insert—often UHMWPE or PTFE-based composite—engineered to pass air but resist fluid. When blood or saline hits it, a swellable or hydrophilic sealing layer activates and closes the vent. That means quick air purge, then an automatic stop. Clinicians like the “no-fuss” factor; biomedical engineers like the predictable sealing curve.
Industry snapshot and trends
Hospitals keep pushing for fewer steps during catheter insertion and better control of blood exposure. Suppliers are responding with tighter pore distributions, faster wet-out shutoff, and cleaner materials (lower extractables). To be honest, the surprising bit is how much customization is happening—ID/OD tuning, airflow targets, and sterilization-ready packaging tailored to OEM assembly lines.
Materials, process, and testing flow
Typical flow: resin selection (medical-grade UHMWPE/PTFE) → powder blending with additives (e.g., hydrophilic or swellable agents) → sintering/molding → precision punching or lathe cut → optional adhesive or press-fit collar → 100% air-flow screening → lot-level bioburden check → packaging for EtO or gamma. Service life is single-use, but shelf life is usually ≈3–5 years (dry storage, away from UV). Real-world use may vary.
Technical specifications (typical)
| Property | Typical Value ≈ | Test/Standard |
|---|---|---|
| Airflow (at 7 kPa) | 50–200 ml/min | ISO 5636 (method adapted) |
| Effective pore size | 5–25 μm | Bubble point (ASTM F316) |
| Sealing response (wet) | ≤1–3 s | Internal functional test |
| Extractables/Leachables | Pass (low) | USP / guidance |
| Biocompatibility | ISO 10993 parts 5/10 | Cytotox./Sensit./Irritation |
| Sterilization compatibility | EtO, Gamma 25–40 kGy | ISO 11135/11137 |
Where it’s used
Primary uses: peripheral IV catheters, blood collection sets, closed-system IV cannulas, oncology infusion lines. Nurses tell me priming feels “more predictable” with a good Iv Catheter Plug, and a few device engineers noted fewer nuisance alarms from residual air pockets during validation.

Vendors at a glance (real-world factors)
| Vendor | Core Material | MOQ ≈ | Customization | Lead Time | Certs (typical) |
|---|---|---|---|---|---|
| China Porous Filters (Shijiazhuang; Rm. C-1301, Hyde Park Plaza, No. 66 Yuhua W. Road, 050056, China) | UHMWPE composite | 10k–50k pcs | ID/OD, airflow, sealing curve | 3–6 weeks | ISO 13485 (facility), ISO 10993 materials |
| Porex | Sintered polymers | 25k+ pcs | High; custom blends | 4–8 weeks | ISO 13485, 10993 data |
| Saint-Gobain | PTFE/ePTFE solutions | Project-based | Engineered assemblies | 6–10 weeks | ISO 13485, sterilization files |
Customization and a quick case
One oncology OEM needed higher initial airflow but a tighter shutoff. The supplier tuned porosity and swapped in a faster-response sealing additive; validation showed air purge time cut by ≈18% while maintaining zero liquid bypass at 10 kPa hydrostatic. Not earth-shattering, but on a line making millions, it matters.
Compliance and validation notes
Look for ISO 13485 QMS, material biocompatibility per ISO 10993 (cytotoxicity, sensitization, irritation), and compatibility with ISO 11135/11137 sterilization. For system-level conformity, infusion devices align with ISO 8536 series. I guess the punchline is simple: tie your Iv Catheter Plug spec to airflow at defined pressure, shutoff time with saline/blood analogs, particulate control (per ISO 8536 or USP guidance), and document it.
Why clinicians and engineers keep it
- Fast priming, fewer accidental drips after wet-out
- Compact form factor; press-fit-friendly
- Consistent sealing without extra user steps
- Scalable manufacturing; cost per unit stays sensible
Citations
- ISO 10993-1:2018, Biological evaluation of medical devices.
- ISO 13485:2016, Medical devices — Quality management systems.
- ISO 8536 series, Infusion equipment for medical use.
- ISO 11135:2014 and ISO 11137-1:2020, Sterilization of health care products.
- USP General Chapters /, Materials of Construction/Polymeric Components (guidance context).
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog