Industry Trends and the Evolution of IV Access Management
The landscape of intravenous (IV) therapy is continually evolving, driven by an imperative for enhanced patient safety, operational efficiency, and cost-effectiveness in healthcare settings globally. Central to this evolution are innovations in IV access devices and accessories. A critical component, often overlooked but vital for maintaining aseptic integrity and preventing complications, is the IV Catheter Plug. Market trends indicate a strong demand for medical devices that offer superior infection control, reduce medication errors, and improve clinician workflow. The global IV solutions market, valued at over $10 billion in 2022, is projected to grow significantly, underscoring the increasing reliance on IV therapies and, consequently, the demand for sophisticated accessory components.
Modern healthcare emphasizes prevention over treatment, pushing manufacturers to develop solutions that minimize risks associated with IV access, such as catheter-related bloodstream infections (CRBSIs). This has led to the development of advanced materials and designs for components like the IV Catheter Plug, focusing on biocompatibility, ease of sterilization, and secure fit. Concurrently, there is a growing interest in devices that integrate passive safety features, requiring minimal active intervention from clinicians to ensure secure connections and occlusions.
Furthermore, the rise of home healthcare and ambulatory care settings necessitates compact, user-friendly, and highly reliable devices. The role of specialized components, including those designed for secure occlusion and, in some cases, features similar to iv catheter vents for managing pressure or air, becomes paramount in these diverse clinical environments.
Technical Specifications of Advanced IV Catheter Plugs
A high-performance IV Catheter Plug is engineered to provide a sterile, leak-proof seal for IV catheters when not in active use. Its design and material composition are critical for maintaining the integrity of the vascular access point and preventing ingress of contaminants. Key technical parameters include material biocompatibility, structural integrity under varying pressures, and ease of aseptic handling.
Product Specification Table: Standard IV Catheter Plug
| Parameter | Specification | Testing Standard |
|---|---|---|
| Material Composition | Medical Grade Polypropylene (PP) / High-Density Polyethylene (HDPE) | USP Class VI, ISO 10993 (Biocompatibility) |
| Luer Lock Compatibility | ISO 594-2 / ISO 80369-7 compliant | Functional dimensional accuracy test |
| Pressure Resistance | Up to 45 psi (310 kPa) without leakage | ISO 8536-12 Annex C (Leakage Test) |
| Sterilization Method | Ethylene Oxide (EO) or Gamma Irradiation | ISO 11135 / ISO 11137 (Sterility Assurance) |
| Packaging | Individual peel-pouch, sterile | ISO 11607 (Packaging Validation) |
| Shelf Life | Minimum 5 years from date of manufacture | Accelerated aging studies per ASTM F1980 |
The precise engineering of the luer taper and thread ensures a secure, twist-on connection, minimizing the risk of accidental dislodgement. Furthermore, certain advanced designs may incorporate features for positive displacement, or even a micro-iv catheter vents system, to manage residual fluid or air, although true vents are less common in simple plugs, which primarily serve as occlusive caps.
Detailed Manufacturing Process Flow for IV Catheter Plugs
The production of a high-quality IV Catheter Plug is a meticulous process, adhering to stringent medical device manufacturing standards. It involves multiple stages, from raw material selection to final sterilization and packaging, ensuring product integrity and patient safety.
Process Steps:
-
Raw Material Sourcing & Qualification:
High-grade medical polymers (e.g., Polypropylene, HDPE) are sourced from approved suppliers. These materials undergo rigorous qualification processes to ensure compliance with USP Class VI and ISO 10993 standards for biocompatibility, chemical inertness, and physical properties. Material traceability is maintained throughout.
-
Injection Molding:
The selected polymers are precisely injected into highly polished molds under controlled temperature and pressure settings. This process forms the body of the IV Catheter Plug, ensuring accurate dimensions and a smooth surface finish. Sophisticated CNC machining is employed in mold fabrication to achieve micron-level precision for the luer lock mechanism.
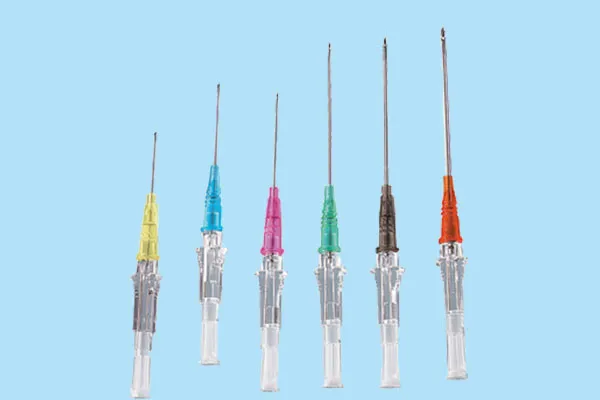
Image 1: Precision injection molding process for IV Catheter Plugs.
-
Automated Assembly & Inspection:
If the plug consists of multiple components, automated assembly lines ensure consistent and sterile integration. High-speed vision inspection systems are utilized to detect any surface defects, dimensional deviations, or particulate contamination. This stage also includes verification of the luer lock fitting as per ISO 80369-7.
-
Functional Testing:
Each batch undergoes rigorous functional testing, including pressure leakage tests (e.g., up to 45 psi), torque retention tests for luer connections, and visual inspection for sealing efficacy. These tests adhere to ISO 8536-12 standards, ensuring a reliable and leak-proof seal.
-
Cleaning & Packaging in Controlled Environments:
The finished components are cleaned in Class 10,000 (ISO Class 7) cleanroom facilities to remove any potential residues. Products are then individually packaged into sterile barrier systems (e.g., peel pouches) in a Class 100,000 (ISO Class 8) cleanroom environment, complying with ISO 11607.
-
Sterilization:
Packaged products undergo validated sterilization processes, typically Ethylene Oxide (EO) or Gamma Irradiation, as per ISO 11135 or ISO 11137 respectively, achieving a Sterility Assurance Level (SAL) of 10-6. Post-sterilization, residual testing for EO is performed if applicable.
-
Quality Control & Release:
Final quality control checks, including sterility testing and batch record review, are performed before products are released for distribution. This ensures full compliance with medical device regulations like FDA 21 CFR Part 820 and ISO 13485.
The stringent adherence to these processes ensures a service life of at least 5 years under proper storage conditions, delivering unparalleled reliability for critical applications in hospitals, clinics, and emergency medical services.
Application Scenarios and Technical Advantages
The versatility and critical function of the IV Catheter Plug make it indispensable across a multitude of healthcare environments. Its primary role is to secure and protect the distal end of an IV catheter or administration set, thereby preventing blood reflux, air embolism, and microbial contamination when the line is not actively in use.
Typical Application Scenarios:
- Intermittent IV Therapy: When patients receive medications or fluids intermittently, the IV Catheter Plug ensures the catheter remains securely sealed between administrations, maintaining sterility and patency.
- Catheter Flushing & Locking: After flushing an IV line, the plug is used to seal the catheter, preventing backflow and maintaining a positive pressure to help prevent occlusions, often replacing older methods like clamping.
- Emergency Medical Services (EMS): In pre-hospital settings, paramedics often establish IV access which may need to be secured and transported before active infusion begins. A robust IV Catheter Plug is crucial for patient safety during transit.
- Home Healthcare: For patients managing IV therapy at home, easy-to-use and highly reliable plugs are essential to minimize infection risk and simplify care routines.
- Operating Rooms & Recovery: Securing IV lines post-procedure or during patient transfer to recovery where active infusion might be paused.
Key Technical Advantages:
- Enhanced Infection Control: Our plugs are designed with a smooth, non-porous surface, minimizing areas for bacterial colonization. The sterile, individual packaging and secure luer lock connection prevent microbial ingress, significantly reducing the risk of CRBSIs, a major healthcare challenge.
- Superior Leakage Prevention: Precision-engineered luer tapers and robust material integrity ensure a leak-proof seal even under moderate internal pressures, safeguarding against blood spills and medication loss. This directly contributes to a cleaner clinical environment and reduced exposure risks for healthcare workers.
- Biocompatibility & Patient Safety: Manufactured from USP Class VI compliant medical-grade materials, our IV Catheter Plug ensures no adverse patient reactions or material degradation within the clinical environment.
- Ease of Use for Clinicians: The ergonomic design facilitates easy and quick application and removal, even with gloved hands, improving workflow efficiency and reducing the potential for user error.
- Cost-Effectiveness: By reducing the incidence of CRBSIs and minimizing the need for re-access due to accidental dislodgement or contamination, these plugs offer significant long-term cost savings associated with extended hospital stays, additional treatments, and material waste.
While primarily designed for occlusion, some specialized accessories in the IV space do act as iv catheter vents to release trapped air, but the core function of a plug remains secure sealing.
Vendor Comparison: Evaluating IV Catheter Plug Solutions
Selecting the right IV Catheter Plug supplier is a strategic decision for healthcare procurement teams, impacting patient safety, clinical efficiency, and operational costs. While many vendors offer similar products, key differentiators lie in material quality, design precision, regulatory compliance, and post-sales support.
Comparative Analysis of Leading IV Catheter Plug Features
| Feature/Vendor | Our Product (HuiBo Filters) | Vendor A (Competitor) | Vendor B (Competitor) |
|---|---|---|---|
| Material Biocompatibility | USP Class VI, ISO 10993 Certified PP/HDPE | USP Class VI PP | Medical Grade PP |
| Luer Lock Standard | ISO 594-2 / ISO 80369-7 | ISO 594-2 | ISO 80369-7 |
| Pressure Resistance (psi) | 45 psi (310 kPa) | 30 psi (207 kPa) | 40 psi (276 kPa) |
| Sterilization Method | EO / Gamma (Validated) | EO | Gamma |
| Manufacturing Certifications | ISO 13485, FDA Registered | ISO 13485 | CE Mark |
| Lead Time (Standard Orders) | 2-4 Weeks | 3-5 Weeks | 4-6 Weeks |
| Customization Options | Extensive (Color, molding, packaging) | Limited (Color) | Moderate (Packaging) |
HuiBo Filters distinguishes itself through its comprehensive adherence to international standards, robust material selection, and proven performance characteristics. Our commitment to continuous improvement and advanced manufacturing techniques ensures that our IV Catheter Plug solutions not only meet but often exceed industry benchmarks for safety and efficacy.
Customized Solutions for Unique Clinical Needs
Recognizing that no two clinical environments or operational protocols are identical, we offer extensive customization capabilities for our IV Catheter Plug range. Our engineering team works closely with clients to develop solutions that precisely integrate with existing systems and address specific challenges.
Customization Avenues Include:
- Material Selection: While medical-grade PP and HDPE are standard, alternative polymers can be explored for specific chemical compatibility requirements or desired tactile properties.
- Color Coding: Custom colors can be implemented for different departments, medication types, or to align with hospital-specific coding systems, enhancing visual identification and reducing the risk of errors.
- Ergonomic Design Modifications: Minor alterations to the grip or cap design can be made to optimize handling for specific user groups or clinical procedures.
- Packaging Configurations: From bulk sterile packaging to custom kit integration, we can adapt packaging to streamline inventory management and clinical workflows. For instance, specific counts or multi-component sterile trays.
- Integrated Features: While typically a passive occlusive device, discussions around incorporating features like integrated lanyards or specialized seals can be explored for unique application requirements. This also extends to integrating features found in iv catheter vents if a combined function is required for specific pressure management.
Our collaborative approach ensures that customized IV Catheter Plug solutions maintain the same high standards of quality, regulatory compliance, and performance as our standard offerings, backed by comprehensive validation and testing.
Application Case Studies
Real-world application demonstrates the tangible benefits of a superior IV Catheter Plug. Our products have been instrumental in improving patient outcomes and operational efficiency across various healthcare partners.
Case Study 1: Large Hospital Network – Reduction in CRBSIs
A major hospital network, comprising five acute care facilities, faced challenges with elevated rates of Catheter-Related Bloodstream Infections (CRBSIs) linked to insecure IV catheter closures. After a comprehensive review, they transitioned to our advanced IV Catheter Plug for all intermittent IV access points. Our plug’s precision luer lock and robust sealing capabilities were critical. Over an 18-month period following implementation, the network reported a 28% reduction in CRBSI rates attributable to IV line management, leading to significant patient safety improvements and an estimated annual savings of over $1.2 million in treatment costs and extended hospital stays.

Image 2: An IV Catheter Plug securely fitted to an IV line.
Case Study 2: Ambulatory Surgical Center – Enhanced Workflow Efficiency
An ambulatory surgical center specializing in same-day procedures sought to streamline its patient preparation and recovery phases. Their previous generic plugs were occasionally difficult to manipulate, particularly for staff wearing multiple layers of gloves, leading to minor delays and frustration. By adopting our ergonomically designed IV Catheter Plug, the center observed a noticeable improvement in application speed and security. Clinician feedback highlighted the easy-grip design and consistent, secure connection. This translated to a 15% reduction in the average time spent on IV line management per patient, contributing to overall operational fluidity and enhanced staff satisfaction.
Frequently Asked Questions (FAQ)
- Q1: What materials are used in your IV Catheter Plug?
- A1: Our IV catheter plugs are primarily manufactured from medical-grade Polypropylene (PP) or High-Density Polyethylene (HDPE), both of which are USP Class VI and ISO 10993 compliant for biocompatibility.
- Q2: Are your IV Catheter Plugs sterile?
- A2: Yes, all our IV Catheter Plug products are supplied sterile, individually packaged in peel pouches, and sterilized via validated Ethylene Oxide (EO) or Gamma Irradiation methods to achieve a Sterility Assurance Level (SAL) of 10-6.
- Q3: What certifications do your products hold?
- A3: Our manufacturing facilities are ISO 13485 certified, and our products comply with international standards such as ISO 594-2, ISO 80369-7, and ISO 10993. We are also FDA registered, ensuring global acceptance and reliability.
- Q4: Can these plugs be used with any luer lock catheter?
- A4: Our IV catheter plugs are universally compatible with all standard male and female luer lock and luer slip connections, adhering strictly to ISO 80369-7 for small-bore connectors for liquids and gases in healthcare applications.
Lead Time, Warranty, and Customer Support
Lead Time & Fulfillment
Standard orders for our IV Catheter Plug typically have a lead time of 2 to 4 weeks, depending on order volume and current production schedules. For customized solutions or large-scale procurement, specific lead times will be provided upon consultation. We maintain robust supply chain management to ensure timely delivery and minimize disruptions for our clients.
Warranty Commitments
We stand behind the quality and performance of our IV Catheter Plug products. All products are guaranteed to be free from defects in material and workmanship for a period of 12 months from the date of shipment, provided they are stored and used according to recommended guidelines. This warranty covers manufacturing defects and ensures compliance with stated specifications.
Dedicated Customer Support
Our commitment to our partners extends beyond product delivery. We offer comprehensive after-sales support, including technical assistance, product training, and responsive handling of any inquiries or concerns. Our dedicated support team is available via phone and email to ensure seamless integration and optimal performance of our IV access solutions in your clinical practice. We pride ourselves on building long-term relationships through unparalleled service and technical expertise.
Contact us today to discuss your specific requirements or to learn more about our range of products.
Authoritative References
- ISO 13485:2016 Medical devices — Quality management systems — Requirements for regulatory purposes. International Organization for Standardization.
- ISO 80369-7:2021 Small-bore connectors for liquids and gases in healthcare applications — Part 7: Connectors for intravascular or hypodermic applications. International Organization for Standardization.
- United States Pharmacopeia and National Formulary (USP-NF) Monographs for Medical Plastics (USP Class VI).
- Centers for Disease Control and Prevention (CDC). Guidelines for the Prevention of Intravascular Catheter-Related Infections, 2017.
- FDA, 21 CFR Part 820: Quality System Regulation. U.S. Food and Drug Administration.
POROYAL is dedicated to the research and development of powder sintered porous filter technology, establishing itself as a leading innovator in advanced porous material solutions.Titanium and plastic filters POROYAL engineers deliver high-value engineering solutions to address complex product design challenges across diverse industries, including petrochemical, oilfield, water treatment, food & beverage, pharmaceutical, chemical processing, medical devices, vacuum conveying, and other specialized applications.Metal porous filter Our expertise encompasses comprehensive liquid-solid and gas-solid separation technologies tailored to meet customer-specific requirements.At POROYAL, we function as an integrated enterprise comprising four specialized divisions—dedicated to the development of Metal Sintered Porous Filters, Plastic Sintered Porous Filters, Metal Fibers, and Medical & Laboratory filtration solutions. Metal Porous Filters Manufacturer
Through collaborative innovation, we engineer sustainable technologies that enhance performance, optimize efficiency, and deliver measurable success for our customers.China Plastic Porous Filtersr|super blog